Novaryn B17 / Mechanism of Action
The Biochemical Pathway of Amygdalin
A mechanistic overview of Amygdalin's enzymatic metabolism, cellular interactions, and the differential response hypothesis.
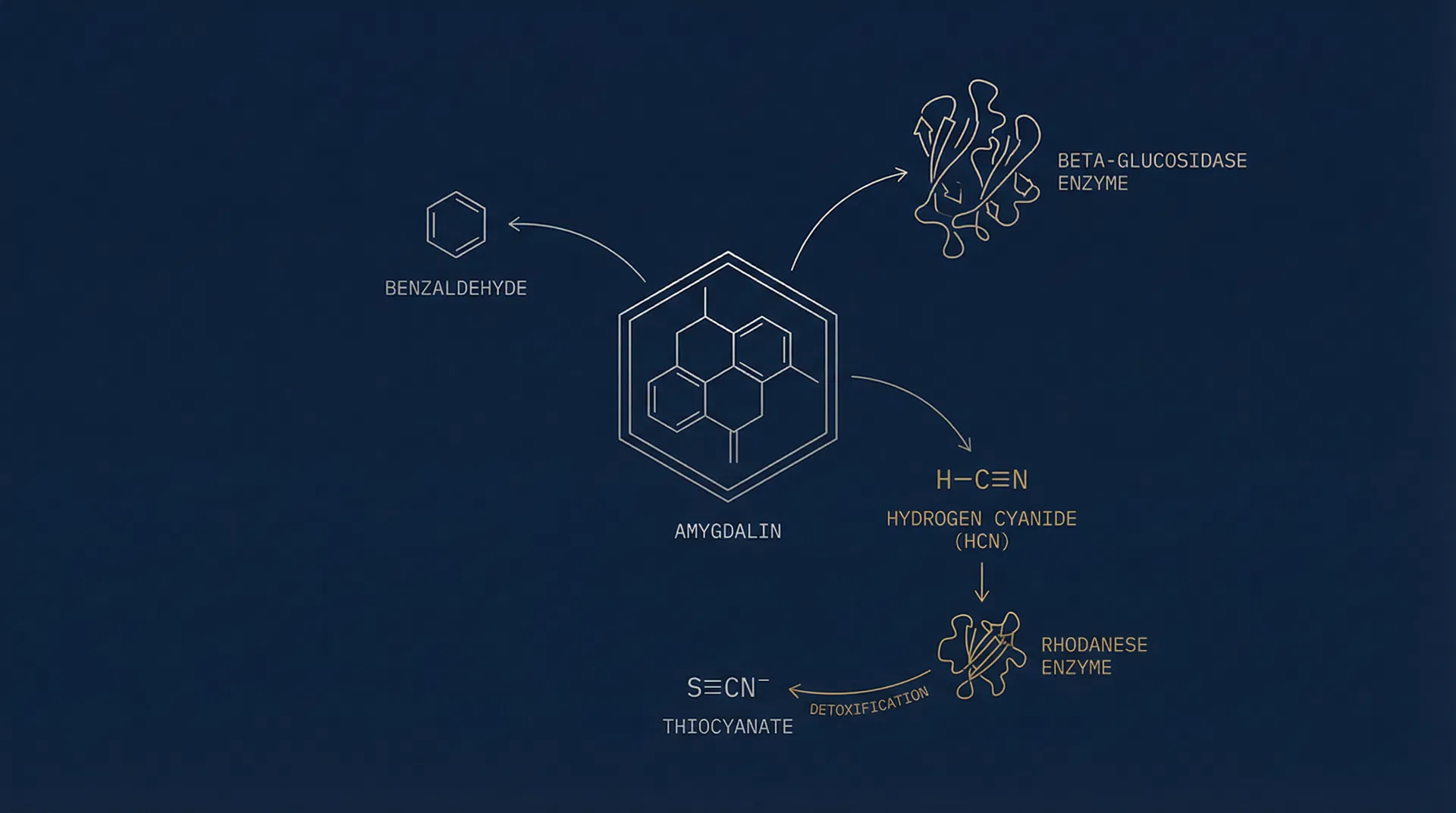
Figure 1. Simplified metabolic pathway of Amygdalin (C₂₀H₂₇NO₁₁) showing key enzymatic interactions.
01 / Overview
Amygdalin: A Cyanogenic Glycoside with a Complex Metabolic Profile
Amygdalin (D-mandelonitrile-β-D-glucosido-6-β-D-glucoside; molecular formula C₂₀H₂₇NO₁₁) is a naturally occurring cyanogenic glycoside isolated primarily from the seeds of Prunus armeniaca (apricot), Prunus amygdalus amara (bitter almond), and related stone fruit species. Its biochemical activity is fundamentally governed by the enzymatic environment it encounters upon administration — a distinction that underpins the selective interaction hypothesis that has driven decades of scientific inquiry.
Unlike simple compounds with direct pharmacological targets, Amygdalin's activity is mediated through a two-enzyme system: beta-glucosidase (BGD), which initiates hydrolysis, and rhodanese (RHD), which governs detoxification. The relative activity of these enzymes within different cell types forms the biochemical basis for the differential cellular response observed in research settings.
02 / Beta-Glucosidase
Hydrolysis via Beta-Glucosidase: The Activation Pathway
The primary metabolic activation of Amygdalin proceeds through the enzyme beta-glucosidase (β-glucosidase; EC 3.2.1.21), a glycoside hydrolase present in intestinal flora, certain plant tissues, and — critically — in varying concentrations across different mammalian cell types. Upon contact with beta-glucosidase, Amygdalin undergoes sequential hydrolysis:
Amygdalin → Prunasin + Glucose
Beta-glucosidase cleaves the outer glucose unit, producing prunasin (D-mandelonitrile-β-D-glucoside) and free glucose.
Prunasin → Mandelonitrile + Glucose
A second hydrolytic cleavage removes the remaining glucose moiety, yielding mandelonitrile.
Mandelonitrile → Benzaldehyde + HCN
Mandelonitrile undergoes spontaneous or enzyme-assisted decomposition, releasing benzaldehyde and hydrogen cyanide (HCN).
The benzaldehyde component has been independently studied for cytotoxic and analgesic properties. The hydrogen cyanide (HCN) component is the primary subject of the selective toxicity hypothesis, as it acts as an inhibitor of cytochrome C oxidase — Complex IV of the mitochondrial electron transport chain — thereby disrupting oxidative phosphorylation and triggering downstream apoptotic signaling.
03 / Rhodanese
Rhodanese: The Detoxification Enzyme
Rhodanese (thiosulfate sulfurtransferase; EC 2.8.1.1) is a ubiquitous mitochondrial enzyme responsible for the primary detoxification of cyanide in mammalian systems. It catalyzes the transfer of a sulfur atom from thiosulfate (S₂O₃²⁻) to cyanide (CN⁻), producing the non-toxic thiocyanate (SCN⁻) and sulfite (SO₃²⁻):
In normal cells, rhodanese activity is sufficient to neutralize HCN produced from Amygdalin metabolism, converting it to thiocyanate — a compound excreted harmlessly via the urinary system. The rate-limiting factor in this process is the availability of thiosulfate as a sulfur donor.
Research has explored the differential expression of rhodanese across cell types, with some studies suggesting reduced rhodanese activity in certain cellular environments — a finding that forms a central component of the selective interaction hypothesis.
04 / Differential Response
The Differential Enzymatic Response Hypothesis
The core mechanistic hypothesis surrounding Amygdalin's cellular selectivity rests on the differential expression of two enzymes: elevated beta-glucosidase activity (increasing HCN production) combined with reduced rhodanese activity (decreasing HCN detoxification) in certain cell populations, compared to the inverse profile observed in normal cells.
Normal Cells
Studied Cell Types
It is important to note that this hypothesis remains an active area of scientific investigation. While in vitro studies have demonstrated differential enzymatic profiles and apoptotic responses, the translation of these findings to in vivo systems and human clinical contexts requires continued rigorous research. Novaryn B17 presents this information for educational and scientific reference purposes only.
05 / Additional Pathways
Beyond the Cyanide Pathway: Additional Mechanisms of Interest
Contemporary research has expanded the mechanistic understanding of Amygdalin beyond the classical HCN pathway. Several additional molecular interactions have been identified in published literature:
Apoptotic Pathway Modulation
Amygdalin has been reported to modulate Bcl-2 (anti-apoptotic) and caspase-3 (pro-apoptotic) gene expression, influencing programmed cell death pathways independently of HCN.
Saleem et al., 2018; PMC10531689
NF-κβ Pathway Inhibition
Studies indicate Amygdalin inhibits nuclear factor kappa-beta (NF-κβ) signaling, a key regulator of inflammatory gene expression, suggesting anti-inflammatory activity.
MDPI Pharmaceuticals, 2021
AMPK/mTOR Pathway
Research has explored Amygdalin's interaction with AMPK/mTOR signaling — a central metabolic regulatory pathway — demonstrating potential for cellular energy homeostasis modulation.
El-Sewedy et al., 2023; PMC10508032
Immunomodulatory Effects
Amygdalin has demonstrated immunomodulatory activity in multiple studies, including effects on T-cell populations, NK cell activity, and cytokine profiles.
Alwan et al., 2022; PMC9816781
Educational Disclaimer
The information presented on this page is for educational purposes only. It summarizes published scientific literature on the biochemical properties of Amygdalin and is not intended as medical advice. It does not constitute a claim that Novaryn B17 products diagnose, treat, cure, or prevent any disease. Consult a qualified healthcare professional before use.